UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM
CURRENT REPORT
Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934
Date of Report (Date of earliest event reported): |
(Exact name of Registrant as Specified in Its Charter)
(State or Other Jurisdiction |
(Commission File Number) |
(IRS Employer |
||
|
|
|
|
|
|
||||
|
||||
(Address of Principal Executive Offices) |
|
(Zip Code) |
||
Registrant’s Telephone Number, Including Area Code: |
(Former Name or Former Address, if Changed Since Last Report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
Securities registered pursuant to Section 12(b) of the Act:
|
|
Trading |
|
|
|
|
|||
|
|
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§ 230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§ 240.12b-2 of this chapter).
Emerging growth company
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act.
Item 7.01 Regulation FD Disclosure.
On September 10, 2021, 23andMe Holding Co. participated in the Wells Fargo Healthcare Conference. The materials attached as Exhibit 99.1 to this Current Report on Form 8-K were distributed to the participants of such conference, which information is incorporated herein by reference.
The information in this Item 7.01 of this Form 8-K and the exhibit attached hereto are being furnished and shall not be deemed “filed” for purposes of Section 18 of the Exchange Act, nor shall they be deemed incorporated by reference in any filing under the Securities Act of 1933, as amended, except as may be expressly set forth by specific reference in such filing.
Item 9.01 Financial Statements and Exhibits.
(d) Exhibits
Exhibit Number |
Description |
|
|
|
|
99.1 |
|
|
|
|
|
104 |
|
Cover Page Interactive Data File - the cover page interactive data file does not appear in the Interactive Data File because its XBRL tags are embedded within the Inline XBRL document |
|
|
|
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned thereunto duly authorized.
|
|
|
|
23ANDME HOLDING CO. |
|
|
|
|
|
Date: |
September 10, 2021 |
|
By: |
/s/ Steven Schoch |
|
|
|
|
Name: Steven Schoch |
|
|
|
|
Chief Financial and Accounting Officer |
|
|
|
|
|

Investor Presentation September 2021 EXHIBIT 99.1

Disclaimer Forward-Looking Statements This presentation contains forward-looking statements within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended, including statements regarding the future performance of 23andMe’s businesses in consumer genetics and therapeutics and the growth and potential of its proprietary research platform. All statements, other than statements of historical fact, included or incorporated in this presentation, including statements regarding 23andMe’s strategy, financial position, funding for continued operations, cash reserves, projected costs, plans, and objectives of management, are forward-looking statements. The words "believes," "anticipates," "estimates," "plans," "expects," "intends," "may," "could," "should," "potential," "likely," "projects," "continue," "will," “schedule,” and "would" or, in each case, their negative or other variations or comparable terminology, are intended to identify forward-looking statements, although not all forward-looking statements contain these identifying words. These forward-looking statements are predictions based on 23andMe’s current expectations and projections about future events and various assumptions. 23andMe cannot guarantee that it will actually achieve the plans, intentions, or expectations disclosed in its forward-looking statements and you should not place undue reliance on 23andMe’s forward-looking statements. These forward-looking statements involve a number of risks, uncertainties (many of which are beyond the control of 23andMe), or other assumptions that may cause actual results or performance to be materially different from those expressed or implied by these forward-looking statements. The forward-looking statements contained herein are also 8-K filed with the Securities and Exchange Commission (“SEC”) on June 21, 2021 and in 23andMe’s Current Report on Form 10-Q filed with the SEC on August 13, 2021, as well as other filings made by 23andMe with the SEC from time to time. Investors are cautioned not to place undue reliance on any such forward-looking statements, which speak only as of the date they are made. Except as required by law, 23andMe does not undertake any obligation to update or revise any forward-looking statements whether as a result of new information, future events, or otherwise. Non-GAAP Financial Measures This presentation also includes references to Adjusted EBITDA, which is a non-GAAP financial measure that 23andMe defines as net income before net interest expense (income), net other expense (income), which includes changes in the fair value of the warrants, depreciation and amortization of fixed assets, amortization of internal use software, non-cash stock-based compensation expense, and expenses related to restructuring and other charges, if applicable for the period. 23andMe evaluates the performance of each segment of its business based on Adjusted EBITDA and has provided a reconciliation of net loss, the most directly comparable GAAP financial measure, to Adjusted EBITDA within this presentation. Adjusted EBITDA is a key measure used by management and the board of directors to understand and evaluate operating performance and trends, to prepare and approve 23andMe’s annual budget and to develop short and long-term operating plans. 23andMe provides Adjusted EBITDA because 23andMe believes it is frequently used by analysts, investors and other interested parties to evaluate companies in its industry and it facilitates comparisons on a consistent basis across reporting periods. Further, 23andMe believes it is helpful in highlighting trends in its operating results because it excludes items that are not indicative of 23andMe’s core operating performance. In particular, management believes that the exclusion of the items eliminated in calculating Adjusted EBITDA provides useful measures for period-to-period comparisons of 23andMe’s business. Accordingly, management believes that Adjusted EBITDA provides useful information in understanding and evaluating operating results in the same manner as 23andMe’s management and board of directors. Adjusted EBITDA has limitations as an analytical tool and you should not consider it in isolation or as a substitute for analysis of 23andMe’s results as reported under GAAP. 23andMe may in the future incur expenses similar to the adjustments in the presentation of Adjusted EBITDA. In particular, 23andMe expects to incur meaningful share-based compensation expense in the future. Other limitations of Adjusted EBITDA include (i) the lack of reflection of capital commitments to be paid in the future, and (ii) although depreciation and amortization are non-cash charges, the underlying assets may need to be replaced, which capital expenditures are not captured by Adjusted EBITDA. 23andMe’s presentation of Adjusted EBITDA should not be construed as an inference that future results will be unaffected by these expenses or any unusual or non-recurring items. Other companies, including companies in the same industry, may calculate Adjusted EBITDA differently or may use other measures to evaluate their performance, all of which could reduce the usefulness of Adjusted EBITDA as a tool for comparison. There are a number of limitations related to the use of Adjusted EBITDA rather than net loss, which is the most directly comparable financial measure calculated in accordance with GAAP. When evaluating 23andMe’s performance, you should consider Adjusted EBITDA alongside other financial performance measures, including net loss and other U.S. GAAP results. Intellectual Property All rights to the trademarks, copyrights, logos and other intellectual property listed herein belong to their respective owners 23andMe’s use thereof does not imply an affiliation with, or endorsement by the owners of such trademarks, copyrights, logos and other intellectual property. Solely for convenience, trademarks and trade names referred to in this Presentation may appear with the ® or ™ symbols, but such references are not intended to indicate, in any way, that such names and logos are trademarks or registered trademarks of 23andMe. Industry and Market Data This Presentation relies on and refers to certain information and statistics based on 23andMe’s management’s estimates, and/or obtained from third party sources which it believes to be reliable. 23andMe has not independently verified the accuracy or completeness of any such third party information.

Behind Every Data Point is a Human Being 1

100,000 Our Mission is to Help People Access, Understand and Benefit from the Human Genome 11.6M 1M 825,000 500,000 366,000 219,000 500,000 REGENERON ALL OF US MILLION VETERAN PROGRAM FINNGEN UK BIOBANK DECODE GENETICS GENOMICS ENGLAND Size and scale of 23andMe enables rapid, novel discoveries

-15 The net positive score Americans gave the pharmaceutical industry 75% Consumers wish their healthcare experience was more personalized 25% U.S. healthcare spending is waste <12% Probability of success for a drug to be approved, taking ~10 years and costing $2.6B to develop 1 The Healthcare System is Dysfunctional 2 3 4 “Of course our system isn’t about healthcare, it’s about maximizing revenue for a whole bunch of different players that have nothing to do with what’s good for patients.” Elisabeth Rosenthal (Editor-in-Chief, Kaiser Health News) 1 JAMA, “Waste in the US Health Care System” (2019). 2 Redpoint Global / Dynata survey of over 1,000 U.S. consumers (2020). 3 Gallup, “Americans’ Views of U.S. Business and Industry Sectors” (2020). 4 PhRMA, “Biopharmaceutical Research & Development: The Process Behind New Medicines” (2015).

Consumer Scale and Empowerment is the Key to Disrupting Healthcare Media Transportation Hospitality Healthcare “Healthcare cannot change from within, it will need an outside force to change it, and that force will be our customers.” Anne Wojcicki Commerce

We Pioneered Digital DTC Healthcare to Empower Customers With Affordable, Direct Access 2015 Carrier Status (inherited conditions) 2016 GHR (genetic health risk) 2018 PGt (pharmacogenetic metabolism) 2019 MUTYH (colorectal cancer) 2017 BRCA (breast and ovarian cancer) 2020 PGt (pharmacogenetic drug response) Proven accuracy (99% NPV/PPV) and accessibility1 TIME MAGAZINE INVENTION OF THE YEAR 6 FDA Authorizations 1 See FDA De Novo Authorizations 140044, 160026, 170046 and 180028 and FDA 510K Clearances K182784 and K193492.

Providing Customers With Key, Actionable Insights “Like me, there are many women who have slipped through the cracks of our current medical screening system, either because they don’t have a family history of breast or ovarian cancer. Or they do not know that they have Ashkenazi Jewish ancestry. In my case, even though I know I have Ashkenazi ancestry, that wasn’t enough to prompt my doctor to consider screening. So there are many women walking around with this risk, who, like me, would have never known of their own risk but for this test from 23andMe.” 23andMe customer who discovered she had a BRCA1 mutation 9,000+ Customers with Hypercholesterolemia (FH) variants 7,000+ Customers with a tested BRCA1 / BRCA2 variant 12,000+ Customers with an increased risk for Chronic Kidney Disease 80% Customers receive a report with a meaningful genetic variant Note: Estimates based on penetrance of variants in 23andMe’s Database.

“The mission of 23andMe is not just about genetics. We want to transform healthcare...What I have learned after 11 years is that people want to participate in research…They don’t want to be a human subject. They want to be respected as an equal and as a partner in the process.” Anne Wojcicki to Recode Decode (2018) Transforming Healthcare With 23andMe’s Crowdsourced, Genetic Database 2

A C G T We are all 99.5% genetically alike ...is a data problem, a very big data problem Unlocking the Genetic Code Creates the Opportunity to Revolutionize the Diagnosis, Prevention and Treatment of Most, if Not All, Human Disease 3 billion base pairs long Cracking the code… 10

We Are Redefining Healthcare. With Data. At Scale. Enabling Research & Services Empowering Consumers 11.6M Genotyped Customers1 4B+ Phenotypic Data Points1 Developing Therapeutics 40+ Program2 Cumulative Genotyped Customers (in M, fiscal year ends March 31) FY17A FY18A FY19A FY20A FY21A FY22Q1 2.0 4.4 7.8 9.8 11.3 11.6 10M+ Genetic Profiles Created Critical Mass 1As of June 30, 2021. 2As of March 31, 2021. Programs include collaborated, 100% owned and royalty interest targets.

Novel Consumer Products Insights Drug Discoveries 40+ Programs2 Phenotypic Data Genetic Data 11.6M Genotyped Customers1 ~65%30K+ Participate in Health Survey3 Innovative Results Return Value to the Customer We run hundreds of billions of association tests per year that further our unique understanding of human biology Consumer Powered Healthcare Flywheel Consumer Research Therapeutics / Product 1As of June 30, 2021. 2As of March 31, 2021. Programs include collaborated, 100% owned and royalty interest targets. 12 Data 80% Opt-In to Research

Our Ancestry Service A Mass Entry Point to Building a Revolutionary Database DNA Relatives Visualize Genetic Connections With an Automatically Built Family Tree Ancestry Composition Note: Opt-in required for DNA Relatives and Family Tree builder.

How Ancestry Matters In Connection To Your Health Ann M. 23andMe Customer Ann did not know her ancestry origins and would not have been eligible for clinical testing under current guidelines. Ann decided to do 23andMe to learn more about her potential health risks. Based on her 23andMe report, she discovered she had a BRCA1 mutation. Her doctor confirmed the results and she opted to have surgeries to reduce her risk of having ovarian and/or breast cancer. Current clinical guidelines and eligibility for insurance coverage limit BRCA testing to women with a personal or family history of cancer (Robson, 2003) Adult individuals with a BRCA1 or BRCA2 variant 50% 50% Meet NCCN® criteria1 Do not meet NCCN® criteria 80% 20% Identified by healthcare system Missed by healthcare system 45% 21% No first-degree family history of a BRCA-related cancer Did not self-report having Jewish ancestry DTC Testing 1 NCCN is the National Comprehensive Cancer Network® (NCCN®). 14

Pharmacogenetics Carrier Status Wellness 30 Including: Type 2 Diabetes (Powered by 23andMe Research) Celiac Disease Uterine Fibroids Chronic Kidney Disease G6PD Deficiency MUTYH-Associated Polyposis BRCA1/BRCA2 (selected variants) Health Predispositions 8 Including: Muscle Composition Genetic Weight Alcohol Flush Reaction Saturated Fat and Weight Sleep Movement 40+ Including: Cystic Fibrosis Sickle Cell Anemia Familial Hyperinsulinism (ABCC8-Related) Tay-Sachs Disease Glycogen Storage Disease (Type 1a) 3 Including: SLCO1B1 Drug Transport CYP2C19 Drug Metabolism e.g., citalopram and clopidogrel DPYD Drug Metabolism Our Health Service The First and Only Multi-Disease DTC Genetic Service That Includes FDA-Authorized Reports and Provides Personalized Genetic Insights and Tools 6 FDA Authorizations 1 1 Wellness information does not require FDA Authorization.

A Meaningful, Engaging (and Fun) Experience Strong Engagement and Trust Drive Longitudinal Data Collection ~80% customers consent to research 4B+ phenotypic data points 180+ published research papers 16

Subscription is the Next Phase of Our D2C Journey Pharmacogenetics 3 reports (FDA-Authorized) Heart Health Reports Atrial Fibrillation, Coronary Artery Disease, LDL Cholesterol, Hypertension DNA Relatives Advanced filters, access up to 5,000 relatives Polygenic Risk Scores (Powered by 23andMe Research) Rapidly discovering new genetic insights: Cancer risk Reproductive Health Diet Sleep Fitness and injuries Migraines Soft Launch October 2020

Genetic Data Helps Drive Behavior Change Eat healthier Set future goals to be healthier Adopt a healthier lifestyle in general Exercise more Get more rest / sleep Stop drinking / drink less Stop smoking / smoke less 76% Report taking a positive health action1 55% 51% 50% 45% 42% 16% 7% 1 Based on 2019 online survey, designed by 23andMe and M/A/R/C Research, of 1,046 23andMe Health + Ancestry customers.

Opportunity for Personalized Healthcare at Scale Practice of Medicine Today Reactive – no customization until symptomatic Proactive – truly individualized from the very beginning

Genetics-Based Primary Care Genetics-Based Approach Will Transform the Continuum of Care 70% Providers think genetic tests will improve clinical outcomes1 Genetics-Based Primary Care Telehealth Diagnostics Testing Genetics-Based Primary Care Wellness Reports Pharmacy / E-Prescribing Medical Records Genetics-Based Primary Care Wearables Hospital Connection 1 Health Affairs, “Views Of Primary Care Providers On Testing Patients For Genetic Risks For Common Chronic Diseases.” (2018).

Transforming Therapeutic Development With the 23andMe Database 3

$2.6B average cost of drug development3 ~90% failure rate2, 3 7 years average time-to-IND1 Drug Development is Inefficient Limited Use of Data and Lack of Patient Engagement Constrain Productivity 1 IND = Investigational New Drug Application. fdareview.org, “The Drug Development and Approval Process” (2020). 2 Probability of success for a drug to be approved is estimated to be <12%. 3 PhRMA, “Biopharmaceutical Research & Development: The Process Behind New Medicines” (2015).

The support of human genetic evidence for approved drug indications Nelson et. al 2015 7 years average time-to-IND1 ~90% failure rate2 years to IND with CD96 drug Targets with genetic evidence have historically had a higher success rate3 23andMe Can Efficiently Develop Novel Therapeutics by Power, Need and Speed NATURE GENETICS PUBLICATION 1 IND = Investigational New Drug Application. fdareview.org, “The Drug Development and Approval Process” (2020). 2 Probability of success for a drug to be approved is estimated to be <12%. PhRMA, “Biopharmaceutical Research & Development: The Process Behind New Medicines” (2015). 3 Nature Genetics Publication, “The support of human genetic evidence for approved drug indications” (2015). ~4 Pharmaceutical Industry 23andMe

Our Scale Enables Real-Time Genetics Health Research1 COVID-19 Research March 16 April 6 June 8 Kicked Off Study Launched Study Preliminary Findings 750K Consumers participated in the COVID-19 study in the first 90 days 1,100,000 COVID-19 study participants Sept. 7 Printed Findings3 (January 2021) 1,876,573 High cholesterol 358,275 Type 2 Diabetes 37,853 Type 1 Diabetes 2,355,068 APOE e4 carriers (Alzheimer’s risk) 1,785,456 Depression 85,604 Epilepsy 1,113,057 Asthma 667,019 Eczema 250,764 Psoriasis 634,734 Irritable Bowel 107,126 UC / Crohn’s 64,800 Barrett’s Esophagus 534,696 Arrhythmia 159,135 Coronary Artery 42,836 Pulmonary Embolism 4,528 Idiopathic Pulmonary Fibrosis 9,047 Systemic Sclerosis 7,334 Sarcoidosis 2 1 As of August 2, 2021. 2 As of January 2021. 3 23andMe COVID-19 manuscript live on MedRXiv September 7, 2020.

GWAS is a statistical analysis of Single Nucleotide Polymorphisms (SNPs), looking to identify differences in frequency between disease cases and controls. SNPs linked with disease will be found at different frequencies in cases versus controls. Association is represented by the level of statistical significance (p-value) of the SNP frequency difference. SNPs can be tested across the genome and mapped to specific regions. Genome-Wide Association Studies (GWAS) GGCCAGCTGGACGAGG GGCCAGCTGGATGAGG SNP Cases Controls

Size and Scale Accelerate Target Discovery 1 Genome-Wide Association Study. Number of independent hits p<5e-8 2015 2016 2017 2018 2019 2020 New programs are identified through GWAS1 hits, which increase as size of database grows 10,000 5,000 0 15,000 20,000 25,000 30,000 35,000 40,000 2016 2017 2021 Example: Number of Osteoarthritis GWAS1 hits dramatically increase as database grows

Cancer Endocrine Hematology Autoimmunity Allergy Infectious Disease Cardiovascular Orthopedic Ophthalmology Neurology G.I. Metabolic Disease NAFLD (Non-Alcoholic Fatty Liver Disease) Hundreds of Distinct Clinical Phenotypes Across Major and Rare Diseases Phenotype Cases Controls Hits New Lost 48048 2517644 104 44 2

10,000’s of Genome-Wide Association Study (GWAS) Hits Determine Disease Associated Genes and Directionality Researching to Understand Compelling Biology Identifying Druggable Proteins Assessment of Unmet Need and Competitive Landscape Best Drug Targets Wet lab validated targets progress through standard stages of research toward the selection of preclinical lead molecules and clinical development Phenotypic Data 23andMe’s database yields thousands of GWAS hits Advanced biology and medicinal chemistry guide design of optimal compounds from initial targets Phenotypic breadth provides unique ability to uncover potential safety issues or possible indication expansions Phenome-Wide Association Studies (PheWAS) Reveal Additional Indications and Potential Safety Concerns Genetic Data Systematic, Scalable Research Platform Yields Novel Drug Targets

Genetic Association of the TSLP Signalling Pathway With Asthma TSLP JAK2 JAK1 STAT5 TSLPR IL-7R𝝰 Proinflammatory signaling Cell membrane TSLP is a well-known cytokine with a role in maintaining immune homeostasis and regulating inflammatory responses at mucosal barriers. The TSLP signaling pathway is an attractive therapeutic target. e.g. Tezepelumab, a TSLP-blocking monoclonal antibody for treatment of asthma. Our genetic data shows that multiple genes within the TSLP pathway associate strongly with asthma.

Breadth of Phenotyping Provides Deeper Genetic Understanding Beyond Single Diseases PheWAS = Phenotype Wide Association Study Every SNP in the genome can be interrogated at >1,000 medically related phenotypes. Besides the role of a gene in a disease of interest, we can use genetics to learn potential indication expansions or possible unwanted toxicities. For TSLP, PheWAS indicates lack of effect in eczema but also highlights potential indication expansion in a rare disease. Rare disease A Asthma

“Our work with 23andMe is exceeding expectations and helping us advance a new way of thinking about drug discovery, one driven by genetics and the DNA we inherit. The insights of why some people are protected from or are at greater risk for certain diseases can lead to genetically validated targets that are at least twice as successful in clinical trials.” Dr. Hal Barron, Chief Scientific Officer & President R&D, GSK (2021) 50/50 shared costs and profits $300M equity investment Access to GSK technology and platforms Strategic Collaboration With

We Have Generated a Deep Pipeline Across Multiple Therapeutic Areas 1 Including GSK unilateral programs. Note: As of March 31, 2021 Immunology Immuno-oncology Phase 1 Data First Time in Human Preclinical Next Milestone Phase 1 Phase 2 Phase 3 Cardiovascular/ Metabolic CD96 P006 Neurology Immuno-oncology EARLY-STAGE THERAPEUTIC AREAS (multiple programs in each area) Gynecology and Infectious Disease 40+ programs1

Our Lead CD96 Program Was Identified With ML and AI Applied to Our Proprietary I/O Genetic Signature Cancer Autoimmune Source: Evaluate Pharma historical and forecast estimates. Large I/O market with over $41B expected in 2021 sales CD96 pathway validated with ML and AI applied to our proprietary I/O genetic signature which also identifies marketed I/O drugs We discovered the signaling pathway has a similar genetic I/O signature 2021 projected sales of leading checkpoint inhibitors I/O genetic signature shows opposing effects on autoimmune and cancer phenotypes CD96 plays an important role in regulating NK and T cell antitumor activity $17.0B KEYTRUDA $7.9B OPDIVO $1.8B YERVOY GSK’608 (anti-CD96) is progressing through a Phase 1 multi-ascending dose trial in patients with advanced solid tumors

Our 23andMe I/O Asset, P006, is a Potent Activator of Human T Cells Suppressed by Tumor Antigen Cancer Immune Immune Cancer Ligand Receptor Signaling protein P006 ligand is strongly expressed in a subset of human tumors Immunohistochemistry for P006 ligand in Small Cell Lung Cancer P006 blocks tumor suppression of T cells and activates immune response P006 expected to enter clinical trials by end of FY2022 P006 pathway has a strong I/O signature unique to the 23andMe database

We Are Rapidly Scaling Our Therapeutics Discovery Efforts Cumulative Targets Through Validation 19 March 31, 2021 March 31, 2020 March 31, 2019 9 5

Financials 4

Strong Financial Foundation to Invest in Future Growth Potential 1 Strong cash position. Cash of $770 million1 supports 23andMe’s plans for significant investment in Therapeutics' portfolio and strategic initiatives 2 Investing in future growth potential. Increased spending on Therapeutics R&D by 66% in Q1’22 compared to the same quarter in the prior year Growing consumer services and genetic / phenotypic database. Balancing growth with profitability in Consumer and Research Services supports additional investment in Therapeutics' efforts 3 1As of June 30, 2021.

Adjusted EBITDA -$143 to -$158 million Net Loss -$210 to -$225 million Revenue $250 to $260 million FY2022 Guidance Strategic Investments in Future Growth Potential

Income Statement and FY2022 Guidance Note: Fiscal year ends March 31. Quarter Ended June 30, Year Ended March 31, FY2022 FY2021 FY2022 Guidance FY2021 (in $M) Amount Amount Amount Amount Revenue $59 $48 $250 - $260 $244 Cost of Revenue 29 26 N/A 127 Gross Profit 31 22 N/A 117 R&D 44 34 N/A 160 S&M 15 11 N/A 43 G&A 13 14 N/A 99 Total Operating Expenses 72 59 N/A 302 Income (Loss) from Operations (42) (37) N/A (185) Interest and Other (Expense) Income (0) 1 N/A 2 Net Income / (Loss) ($42) ($36) ($225) - ($210) ($184) Adjusted EBITDA (Consolidated) ($27) ($20) ($158) - ($143) ($77)

Revenue Composition Quarter Ended June 30, Year Ended March 31, FY2022 FY2021 FY2021 (in $M, except percentages) Amount Percentage of Revenue Amount Percentage of Revenue Amount Percentage of Revenue Consumer Services $48 81% $35 72% $198 81% Research Services $11 19% $13 28% $46 19% Therapeutics $0 0% $0 0% $0 0% Total $59 100% $48 100% $244 100%
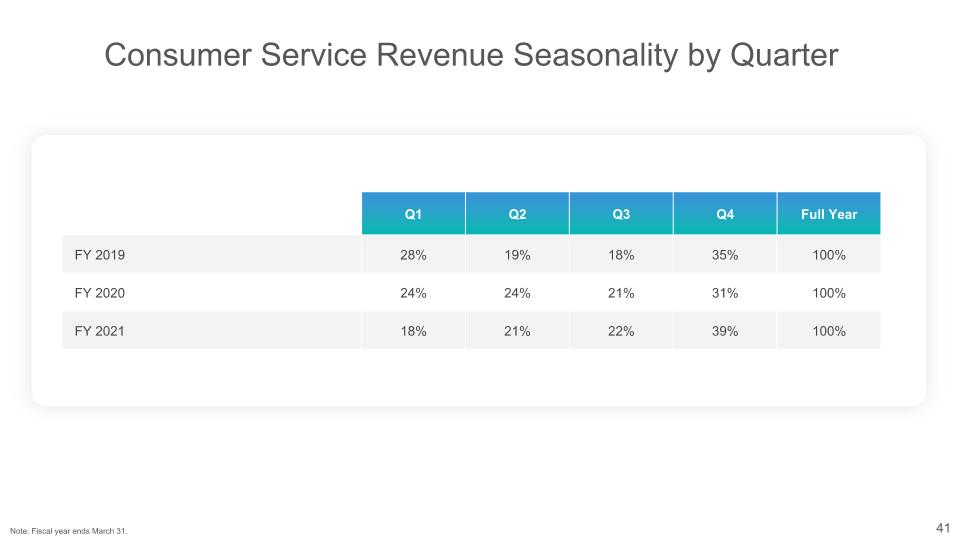
Consumer Service Revenue Seasonality by Quarter Q1 Q2 Q3 Q4 Full Year FY 2019 28% 19% 18% 35% 100% FY 2020 24% 24% 21% 31% 100% FY 2021 18% 21% 22% 39% 100% Note: Fiscal year ends March 31.

Research and Development Expense Quarter Ended June 30, YoY FY2022 FY2021 (in $M, except percentages) Amount Percentage of total R&D expense Amount Percentage of total R&D expense % Change Therapeutics $21 47% $13 37% 66% Consumer and Research Services $23 53% $21 63% 7% Total R&D Expense $44 $34 Investing in Therapeutics

Sales and Marketing Expense Composition Quarter Ended June 30, FY2022 FY2021 (in $M) Amount Amount Advertising and brand $9 $4 Personnel-related expenses $3 $4 Outside Services, equipment and supplies $1 $1 Facilities and other overhead allocation $2 $2 Total $15 $11

Quarter Ended June 30, (unaudited) FY2022 FY2021 (in $K) Amount Amount Segment Revenue Consumer & Research Services $59,239 $48,009 Therapeutics - $48 Total Revenue $59,239 $48,057 Segment Adjusted EBITDA Consumer & Research Services ($505) ($4,236) Therapeutics ($18,303) ($9,394) Unallocated Corporate ($8,467) ($6,199) Total Adjusted EBITDA ($27,275) ($19,829) Reconciliation of Net Loss to Adjusted EBITDA Net Loss ($42,026) ($35,770) Adjustments: Interest (income), net ($44) ($74) Other (income) expense, net $520 ($878) Depreciation and Amortization $4,638 $5,532 Stock-based compensation expense $9,637 $11,361 Total Adjusted EBITDA ($27,275) ($19,829) Note: Fiscal year ends March 31. Segment Information and Reconciliation of Non-GAAP Financial Measures

Outlook for the Year Ending March 31, 2022 (unaudited) Low High (in $M) Amount Amount Reconciliation of Net Loss to Adjusted EBITDA Net Loss ($225) ($210) Adjustments: Interest (income), net ($0) ($0) Other (income) expense, net $1 $1 Depreciation and Amortization $19 $19 Stock-based compensation expense $47 $47 Total Adjusted EBITDA ($158) ($143) Note: Fiscal year ends March 31. Reconciliation of GAAP Net Income Outlook to Non-GAAP Adjusted EBITDA Outlook

FDA Authorized 23andMe+ 125K+ Subscribers2 6 FDA Authorizations Strong Cash Position $770M1 Enabling Research & Services Empowering Consumers 11.6M Genotyped Customers1 4B+ Phenotypic Data Points1 Developing Therapeutics 40+ Programs2 1As of June 30, 2021. 2As of March 31, 2021. Programs include collaborated, 100% owned and royalty interest targets. We Are Redefining Healthcare. With Data. At Scale.

APPENDIX

Imputation Allows Us to Make the Vast Majority of GWAS Discoveries at a Fraction of the Cost of Sequencing Genetic variants are correlated with each other. Knowing the alleles an individual carries at a given position in their genome allows alleles at nearby locations to be inferred. This inference process is known as ‘genotype imputation’. We type ~650,000 SNPs using our genotyping array, which allows accurate imputation for > 35m SNPs in the genome. Genotype imputation is much more cost effective than whole-genome sequencing. Whole-genome sequencing ~$1000 / sample. Exome sequencing ~$400 / sample. Imputation < $0.01 / sample We can impute variants down to ~0.5% frequency, which covers the range at which even large GWAS are statistically powered. We do deploy sequencing in situations where there is a clear benefit over and above imputation. E.g. Rare diseases, founder populations, non-European populations, complex regions of the genome, etc. Before imputation After imputation

23andMe’s Value Proposition 1 Disrupting the Healthcare experience. 23andMe is building a personalized health and wellness experience that caters uniquely to the individual by harnessing the power of their DNA 2 The world’s premier re-contactable phenotype-linked genetic database. A vast (>11M genotyped customers) proprietary dataset rich with both genotypic and phenotypic (health) information allows insights that unlock revenue streams across digital health, therapeutics, and much more Over 40 identified therapeutics programs validates the approach of developing novel therapeutics using genetic data. One program in clinical development with GSK, one wholly owned program expected to start clinical trials before end of March 2022. 4 Continuously increasing quantity and quality of phenotypic data. Impressive customer participation provides >4 billion phenotypic data points for unprecedented statistical power to discover new insights into health and potential therapies. 3 6 Strong cash position. Strong balance sheet supports 23andMe’s plans for significant investment in therapeutics portfolio and strategic initiatives 5 Difficult to replicate platform for value creation. The FDA-approved consumer platform, the therapeutics efforts, and the rich database combine to create multiple opportunities for substantial value creation
